National Certification System for Tissue Culture raised plants
National Certification System for Tissue Culture raised plants
Plant tissue culture technology has played a pivotal role in revolutionizing the growth of agriculture worldwide by large scale clonal multiplication of various economically important plant species. The Department of Biotechnology (DBT), Government of India in 2005 evolved a National Certification System for Tissue Culture Raised Plants (NCS-TCP), in consultation with Ministry of Agriculture and Farmers' Welfare, to provide support to the tissue culture industry for propagation and distribution of virus-free and quality tissue culture raised plants to growers/farmers.
DBT was notified as the Certification Agency for certification of tissue culture raised plants vide Gazette of India Notification No. F. No. 18-28/202-SD.IV dated March 10, 2006 under Section 8 of the Seeds Act, 1966 (54 of 1966) issued by the Ministry of Agriculture and Farmers' Welfare. Gradually, NCS-TCP has become National level mandated activity for tissue culture industry sector in India.
Recognition/Renewal of Recognition of Tissue Culture Production Facilities
Eligibility Criteria for Recognition of Tissue Culture Production Facilities
- Any Tissue Culture Production Facility (Public sector, Private sector, NGO and others) engaged in production of Tissue Culture Plants with minimum production capacity of 0.5 million (5 Lakhs) plants per annum.
- Tissue culture production facility should be fully operational (including all areas of laboratory and hardening facility) at the time of application and site visit.
Process of Recognition/Renewal of Recognition of Tissue Culture Production Facilities (TCPFs)
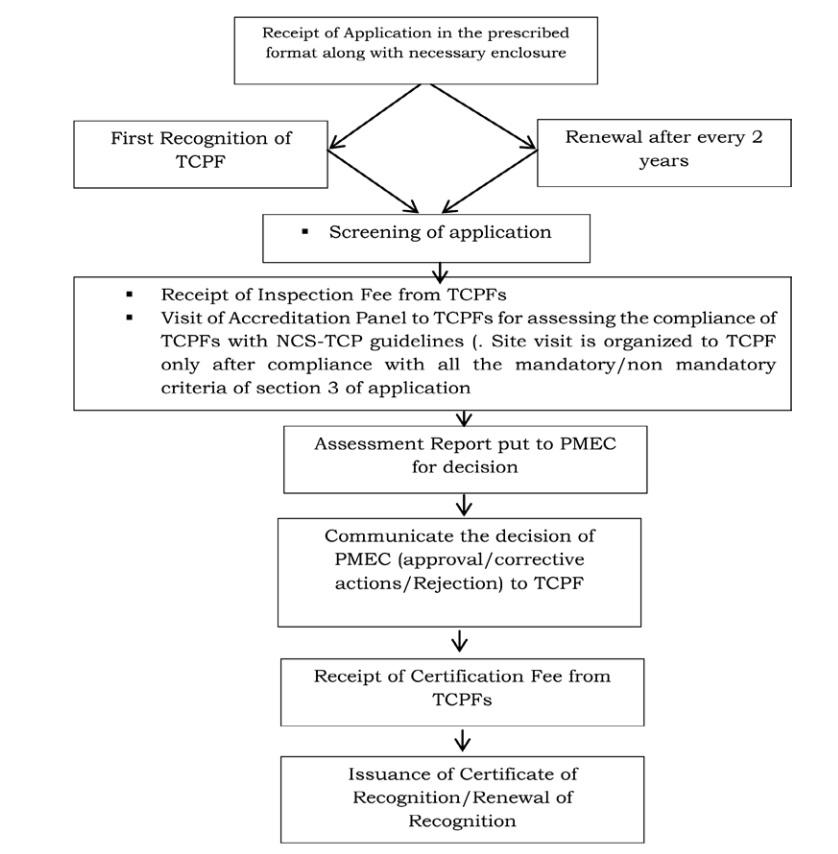
Application form for Recognition/Renewal of Recognition is available at www.dbtncstcp.nic.in. The NCS-TCP Management Cell (NMC) would adhere to time frame for site visit of Accreditation Panel for Recognition/ Renewal of Recognition of TCPFs i.e 30 working days from the time of complete application from TCPFs. In case of non-conformities, verification visit would be organized within30 working days day after formal communication from TCPFs of their corrective actions and receipt of verification visit fee.
Renewal of Recognition: Noteworthy facts
- Application for renewal of Recognition should ideally be submitted 3 months prior to the date of expiry of the “Certificate of Recognition”. Renewal fee registration fee of Rs. 250/- (Rupees two hundred and fifty only) to be deposited in NCS-TCP centralized account mentioned in the application form and particulars of transfer to be submitted along with application form
- NMC will not be responsible for any delay in the renewal of Recognition, if complete application has not been submitted by TCPFs 3 months prior to expiry of Certificate
- If the complete Application of Renewal (NCS-TCP Form- 2A) is not received 1 month prior to the expiry of their “Certificate of Recognition” the company will be asked apply for fresh Recognition in NCS-TCP Form-2.
- Renewal of Recognition would be granted to those TCPFs who are undertaking Certification of every batch of tissue culture raised plants
Certification of Tissue Culture Plants
Eligibility Criteria for Certification of Tissue Culture Raised Plants
The Tissue Culture Production Facility (TCPF) must be Recognized under NCS- TCP. Accredited Test Laboratories (ATLs) would accept samples for certification only from Recognized TCPFs
Requirements for Certification of Tissue Culture Raised Plants
- The mother plant tissue/stock culture must be tested for freedom from all known viruses from ATLs or any reputed Government institutions. To get the list of ATLs established under NCS-TCP, click here.
- The respective batch (lot) of tissue culture plants should be derived from tested stock culture as stated above.
- Recognized TCPFs should assign 4 digits batch number to the said batch of tissue culture plants. This above number should be provided to ATLs while sending samples for certification.
- Tissue culture raised plants ready to dispatch to the farmers (ideally secondary hardened) will be tested for all known viruses and true to type in order to certify under NCS-TCP
Procedures and relevant forms to be used for “Batch Certification of Tissue Culture Raised Plants”
- The Recognized TCPF should sent an intimation form (Virus/ genetic fidelity) Testing for Batch Certification of Tissue Culture Raised Plants to the ATL in a specified format given at https://dbtncstcp.nic.in/Download-Application-Forms
- The ATL would send an Acknowledgement to the TCPF providing details of requisite fee to send the Application form/samples.
- On receipt of acknowledgement, sample would be drawn as per the sampling strategy given below (samples would be drawn either by representative of ATL or TCPF itself as per the mutual consent and coordination between TCPF and ATL)
- The requisite fee to be deposited at centralized NCS-TCP account mentioned in the application form.
- Samples for certification would be sent to ATL along with the duly filled Application for Testing for Batch Certification of Tissue Culture Raised Plants along with particulars of transfer. This application form can be downloaded from the link - http://www.dbtncstcp.nic.in/html/ content/ANNEXURE4.html
For more information, click here.
Source : National Certification System for Tissue Culture Raised Plants (NCS-TCP)
Last Modified : 7/3/2023
